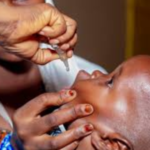
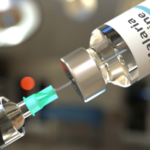
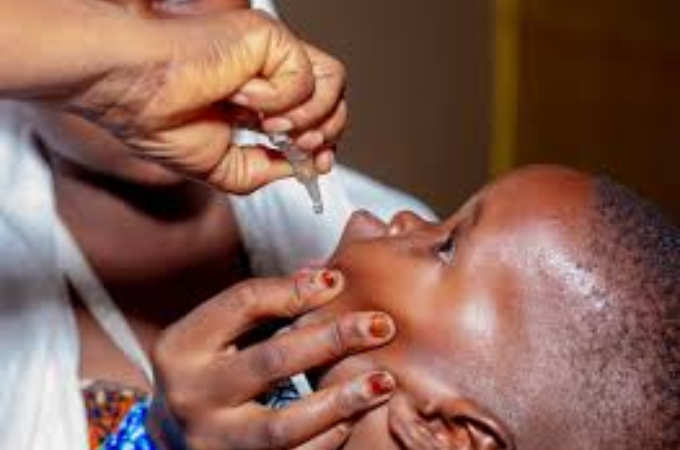
The World Health Organization has granted prequalification status to a new version of the novel oral polio vaccine type 2 (nOPV2), expanding the global supply of a critical tool in the fight against poliovirus type 2 outbreaks and advancing progress toward complete polio eradication.
Prequalification confirms that the vaccine meets WHO’s strict international standards for quality, safety, and effectiveness, allowing it to be procured through UN agencies like UNICEF and used in immunization programs worldwide to prevent and stop poliovirus transmission.
The newly approved product is manufactured by Biological E. Limited in India, using in-house bulk vaccine produced through a technology transfer from PT Bio Farma in Indonesia. This approval adds to earlier WHO listings of nOPV2 made by PT Bio Farma and BioE-formulated versions using PT Bio Farma bulk, further diversifying and strengthening the reliable supply chain for this essential vaccine.
The nOPV2 was specifically developed to tackle outbreaks of circulating vaccine-derived poliovirus type 2 (cVDPV2), which can emerge in communities with low immunization coverage. Unlike traditional monovalent OPV type 2, nOPV2 is genetically more stable, greatly reducing the chance of it seeding new outbreaks while remaining highly effective at quickly halting virus spread.
WHO Director-General Dr Tedros Adhanom Ghebreyesus highlighted the impact of vaccination efforts during his opening remarks at the 158th session of the WHO Executive Board (February 2–7, 2026): “Vaccines are bringing us closer to the eradication of polio, with 41 cases of wild polio reported last year from just 24 districts in Pakistan and Afghanistan, down from 99 cases in 49 districts in 2024.”
Since its introduction, nOPV2 has been deployed in numerous outbreak responses, reaching hundreds of millions of children and helping to reduce cVDPV2 transmission in several affected countries. The vaccine is approved for emergency use in all age groups during type 2 poliovirus outbreaks, as directed by the Global Polio Eradication Initiative or WHO.
Technical Details of the New Product
The BioE-manufactured nOPV2 is supplied in 20-dose and 50-dose vial presentations. It has a 24-month shelf life when stored at or below –20°C and can also be kept for up to six months at +2°C to +8°C, offering important flexibility for immunization programs in diverse settings. Under WHO’s Multi-dose Vial Policy, opened vials can be used for up to 28 days, helping maximize efficiency during outbreak campaigns.
Executive Board Discussions on Poliomyelitis
During the 158th session of the WHO Executive Board, delegates stressed the need to sustain high-quality surveillance, vaccination coverage, and rapid outbreak response in the remaining endemic areas (Pakistan and Afghanistan), while addressing circulating vaccine-derived outbreaks elsewhere. Discussions emphasized that political commitment, access to communities, predictable financing, and strong cross-border coordination will be essential to convert recent progress into permanent eradication and secure a polio-free future.












































































































































































































































































































































![MMIA Fire: Keyamo Inspects Lagos Airport After Terminal 1 Blaze Disrupts Flights Hon. Festus Keyamo SAN arrives MMIA past 11:50 p.m. Feb 23 to lead oversight of Terminal 1 fire. Joined FAAN MD & team actively managing situation. Directed morning inspection Tuesday to guide restoration & confirm safety standards. Reaffirmed focus on passenger safety. Story: [link] #KeyamoMMIA #FireIncident #FAAN](https://fullcliphq.com/wp-content/uploads/2026/02/mym-150x150.jpg)