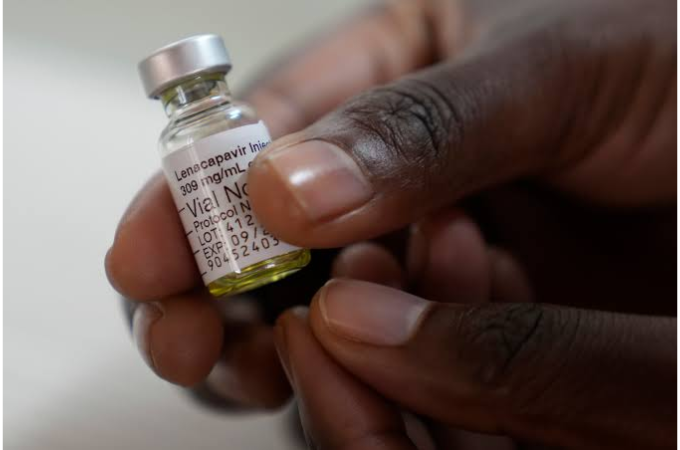
Kenya’s Ministry of Health has announced plans to roll out 21,000 starter doses of lenacapavir – a twice-yearly injectable HIV pre-exposure prophylaxis (PrEP) in early March, positioning the country for a phased national rollout.
Lenacapavir delivers six months of protection per subcutaneous injection for HIV-negative individuals at substantial risk, blocking viral replication stages to prevent infection. It offers advantages in adherence over daily oral PrEP, especially for populations facing barriers like stigma or pill fatigue, though it requires ongoing HIV testing and is not a treatment, cure, or vaccine.
Kenya’s approach stands out for its structured, data-driven phasing: initial coverage in 15 high-prevalence counties (Mombasa, Kilifi, Machakos, Nairobi, Kajiado, Nakuru, Uasin Gishu, Kakamega, Busia, Siaya, Kisumu, Migori, Homa Bay, Kisii, Kiambu), followed by another 15, then the remaining 17 for nationwide reach. This builds on existing PrEP programs through NASCOP, with integration into KEMSA supply chains, strengthened safety monitoring, and a long-term resource plan beyond initial Global Fund support (at ~KSh 7,800 per person/year). Additional doses include 12,000 continuations by April and 25,000 pledged by the U.S. government.
Compared to other early-adopter African countries selected alongside Kenya in July 2025 (Eswatini, Lesotho, Mozambique, Nigeria, South Africa, Uganda, Zambia, Zimbabwe), progress varies significantly as of mid-February 2026.
Eswatini and Zambia moved fastest: both received initial shipments (around 500 doses each) in November 2025 via PEPFAR and Global Fund support, with programmatic administration starting shortly after – often cited as the quickest PrEP product introduction ever, just months post-U.S. FDA approval (June 2025). Eswatini launched in select clinics by World AIDS Day 2025, emphasizing rapid community access.
South Africa secured regulatory approval in October 2025 (first in Africa via SAHPRA), followed by implementation studies through partners like Wits RHI, with broader programmatic rollout planned for early 2026. However, political tensions with U.S. donors led to exclusions from certain PEPFAR-supplied doses, shifting reliance to other funding streams.
Zimbabwe gained approval in November 2025, while regulatory clearances continued in Mozambique (January 2026), Namibia (January 2026), and others. Lesotho, Uganda, and Nigeria remain in preparatory or regulatory stages, with expected arrivals in early 2026 through Global Fund/PEPFAR pipelines – though specific launch dates lag behind Eswatini/Zambia’s early start.
Kenya’s March timeline aligns with the “early 2026” window for many peers but emphasizes a county-level, high-burden focus and domestic readiness (PPB registration January 2026). Its initial volume (21,000 starter + incoming continuations) targets immediate impact in priority areas, contrasting smaller pilot shipments in Eswatini/Zambia or study-focused starts in South Africa.
Across the region, lenacapavir’s introduction accelerates compared to prior PrEP options like cabotegravir (injectable, every two months), which faced slower national scale-up despite earlier approvals. Generic versions from partners (e.g., Dr. Reddy’s, Hetero) aim for $40/person/year availability by 2027, potentially easing costs further.
Kenya’s commitment reflects strong leadership in adopting modern tools amid 3.7% prevalence and youth-heavy new infections (41% under 24). The Ministry stresses equitable integration, community engagement, and monitoring to maximize safety and reach.





































































































































































































































![MMIA Fire: Keyamo Inspects Lagos Airport After Terminal 1 Blaze Disrupts Flights Hon. Festus Keyamo SAN arrives MMIA past 11:50 p.m. Feb 23 to lead oversight of Terminal 1 fire. Joined FAAN MD & team actively managing situation. Directed morning inspection Tuesday to guide restoration & confirm safety standards. Reaffirmed focus on passenger safety. Story: [link] #KeyamoMMIA #FireIncident #FAAN](https://fullcliphq.com/wp-content/uploads/2026/02/mym-150x150.jpg)